Properties of Water
Bonds
Water is a compound. It contains Hydrogen and Oxygen in a fixed proportion, H2O. The Hydrogen and Oxygen are held together by chemical bonds; specifically, covalent bonds, which means the electrons are shared. Water is a polar molecule due to a slightly negative charge on the oxygen and slightly positive charge on the hydrogens so the molecule has a bent shape. This polarity is what allows water to attract other molecules. This property, called cohesion, allows water molecules to stick to each other. Water also tends to stick to other materials, a property called adhesion.
Specific Heat
Water has a high specific heat: 4.186 J/g*C. This means that it takes 4.186 Joules of heat energy to raise the temperature of one gram of water one degree Celsius. Compare this to copper (specific heat=0.386) or glass (specific heat=0.84). This high specific heat means water resists changing temperature (higher or lower) and is what helps regulate climate across the world.
Thermostatic Effects
Thermostatic properties are those that act to moderate changes in temperature.
Differences in hottest & coolest places:
Los Angeles Columbia
Latitude: 34.3 N 34 N
Avg Winter Temp 50-70 °F 30-60 °F
Avg Summer Temp 60-85 °F 70-90 °F
Avg Rainfall 2-3 inches/month 47 inches
Why the difference?? Air masses controlling LA's climate are coming off the ocean (more temperate). Our climate is controlled by air masses coming across land; therefore, they are more likely to change dramatically and are not as mild. (larger weather ranges)
These thermostatic effects affect climate (otherwise the poles would freeze and the equatorial oceans would boil!). As water moves around the ocean warm, tropical water is carried towards the poles and cool, polar water is brought towards the equator.
Water is a compound. It contains Hydrogen and Oxygen in a fixed proportion, H2O. The Hydrogen and Oxygen are held together by chemical bonds; specifically, covalent bonds, which means the electrons are shared. Water is a polar molecule due to a slightly negative charge on the oxygen and slightly positive charge on the hydrogens so the molecule has a bent shape. This polarity is what allows water to attract other molecules. This property, called cohesion, allows water molecules to stick to each other. Water also tends to stick to other materials, a property called adhesion.
Specific Heat
Water has a high specific heat: 4.186 J/g*C. This means that it takes 4.186 Joules of heat energy to raise the temperature of one gram of water one degree Celsius. Compare this to copper (specific heat=0.386) or glass (specific heat=0.84). This high specific heat means water resists changing temperature (higher or lower) and is what helps regulate climate across the world.
Thermostatic Effects
Thermostatic properties are those that act to moderate changes in temperature.
Differences in hottest & coolest places:
- On land = 140 °C
- In water = 34 °C
Los Angeles Columbia
Latitude: 34.3 N 34 N
Avg Winter Temp 50-70 °F 30-60 °F
Avg Summer Temp 60-85 °F 70-90 °F
Avg Rainfall 2-3 inches/month 47 inches
Why the difference?? Air masses controlling LA's climate are coming off the ocean (more temperate). Our climate is controlled by air masses coming across land; therefore, they are more likely to change dramatically and are not as mild. (larger weather ranges)
These thermostatic effects affect climate (otherwise the poles would freeze and the equatorial oceans would boil!). As water moves around the ocean warm, tropical water is carried towards the poles and cool, polar water is brought towards the equator.
Seawater
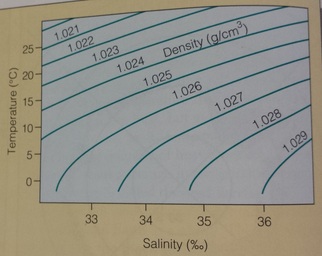
"Seawater is about 96.5% pure water and 3.5% dissolved solids and gases" [Oceanography, Garrison]. These solids ("salts") come from 2 places: chemical weathering of rocks on land which are carried to the ocean by rivers, and from Earth's interior--being released through hydrothermal vents or volcanoes. There are 6 main ions that dissolve: Cl, Mg, K, Ca, Na, and SO4-. Pure water has a density of 1.00 at 3.98 °C. The addition of solids causes an increase in the density to about 1.02 or 1.03.
Salinity (amount of dissolved solids present) .
Pure water is 0% while the ocean is 3.5% These dissolved solids act like an antifreeze. It lowers the freezing point. The freezing point of seawater is -1.91 °C. When seawater freezes, the ice is pure water--> the salts are left out making the surrounding water more salty & denser. This denser water sinks. When seawater evaporates, only the water evaporates leaving behind salts--> saltier water--> more dense--> sinks.
There is an inverse relationship between temperature and salinity. As temperature decreases, salinity increases and therefore density increases. This means that cold seawater is denser than warm seawater.
Water is unique in that it expands when it freezes causing it to become lighter/less dense as a solid. This is why ice floats--if this didn't happen, bodies of water would freeze from the bottom up and eventually there would be no life in the ocean!
***Note that two samples of water can have the same density at different combinations of temperature and salinity***
Interesting Salinities: (remember, most seawater is 3.5%)
Red Sea= 4%
Baltic Sea= 0.7%
Dead Sea= 15-30%
Salinities greatly affect what types of organisms are capable of living there.
Salinity Trends:
Tropics: warmer water due to more direct sunlight but not a lot of changes in water temperature; more evaporation than precipitation so salts get left behind--> higher salinity
Temperate: Temperature varies more; m ore precipitation than evaporation (but not too much)-->Salinity stays pretty stable
Polar: Cooler here but no big changes in air temperature; more precipitation than evaporation; adds more water but not salt so salinity is lower
Salinity (amount of dissolved solids present) .
Pure water is 0% while the ocean is 3.5% These dissolved solids act like an antifreeze. It lowers the freezing point. The freezing point of seawater is -1.91 °C. When seawater freezes, the ice is pure water--> the salts are left out making the surrounding water more salty & denser. This denser water sinks. When seawater evaporates, only the water evaporates leaving behind salts--> saltier water--> more dense--> sinks.
There is an inverse relationship between temperature and salinity. As temperature decreases, salinity increases and therefore density increases. This means that cold seawater is denser than warm seawater.
Water is unique in that it expands when it freezes causing it to become lighter/less dense as a solid. This is why ice floats--if this didn't happen, bodies of water would freeze from the bottom up and eventually there would be no life in the ocean!
***Note that two samples of water can have the same density at different combinations of temperature and salinity***
Interesting Salinities: (remember, most seawater is 3.5%)
Red Sea= 4%
Baltic Sea= 0.7%
Dead Sea= 15-30%
Salinities greatly affect what types of organisms are capable of living there.
Salinity Trends:
Tropics: warmer water due to more direct sunlight but not a lot of changes in water temperature; more evaporation than precipitation so salts get left behind--> higher salinity
Temperate: Temperature varies more; m ore precipitation than evaporation (but not too much)-->Salinity stays pretty stable
Polar: Cooler here but no big changes in air temperature; more precipitation than evaporation; adds more water but not salt so salinity is lower
